Broad GLP-1 availability depletes treatment-naive pools and drives placebo-arm dropout, as patients who experience weight regain abandon trials for commercial alternatives. Investigators hesitate to enroll in placebo-controlled designs when effective options are commercially available. Screen failure rates rise as washout-required candidates refuse to pause therapies that are visibly working.
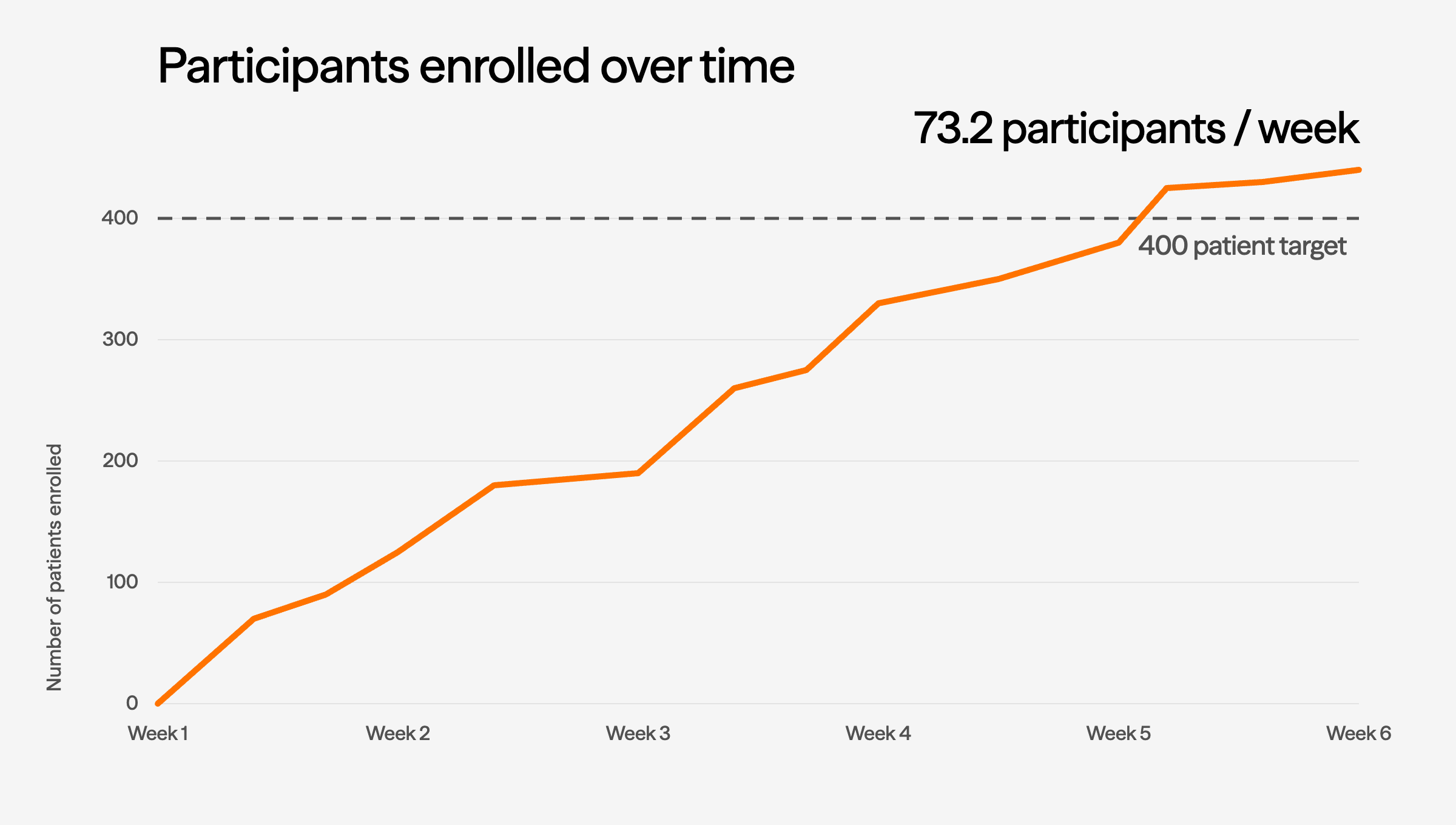
Lindus queries 40M+ EHR records by diagnosis codes, medication history, and treatment gaps to identify candidates before site activation. Patient concierge coordinators sustain placebo-arm retention through scheduled touchpoints, transport, and direct communication: reducing the dropout-to-commercial-therapy pattern that erodes your control arm.