Engineered to deliver your trial on time from startup to readout.
Every function. One system. One team.



Lindus is an Accountable Research Organization built on an AI-native operating system that puts you back in control of your trial timeline.
Real-time visibility
Sponsors and Lindus teams act on the same signal at the same time. Risks surface early enough to act on, not late enough to explain.
Performance-based commercial model
Our incentives sit exactly where yours do: on predictability and control over your timelines.
Integrated operating system
One execution path from design through database lock. No handoffs. No accountability gaps. Unparalleled flexibility to reach the right patients and sites, and to overcome operational roadblocks.
Citrus™ - AI-native operating system
One AI-native operating system, Citrus™, connects everything from patient identification to final data capture. Integrated workflows surface risk early enough to act, across every function.
Centralized patient identification and enrollment
Centralized patient enrollment infrastructure designed to identify eligible patients early, screen and enroll them efficiently. By combining AI-powered EHR querying, digital pre-screening, and centralized patient support, we identify patients directly within real-world care pathways and guide them through screening and site enrollment. This approach reduces screen failures, accelerates enrollment timelines, and removes recruitment burden from sites.

Eligible patients are identified directly within real-world healthcare systems before traditional site recruitment begins. Multi-channel outreach reduces dependence on site referral networks when recruitment slows.
Automated screening of patient records against protocol eligibility criteria at scale reduces the manual burden of candidate identification and cuts screen failure rates significantly.
Dedicated coordinators support patients through scheduling, education, and site connection — minimizing the drop-off that commonly occurs between identification and first study visit.
Flexible site models
A site model built around what your trial needs: fully site-based, hybrid, or fully virtual, all running through the same integrated system with the same team and the same standards.
Integrated clinical services
Clinical trial services delivered by one team accountable to operational performance, from protocol design through database lock. AI-enabled feasibility assessment draws on 40 million real-world EHR records to model enrollment trajectories, run scenario analyses against eligibility criteria, and stress-test design assumptions. Bottlenecks are identified before any sites are activated.

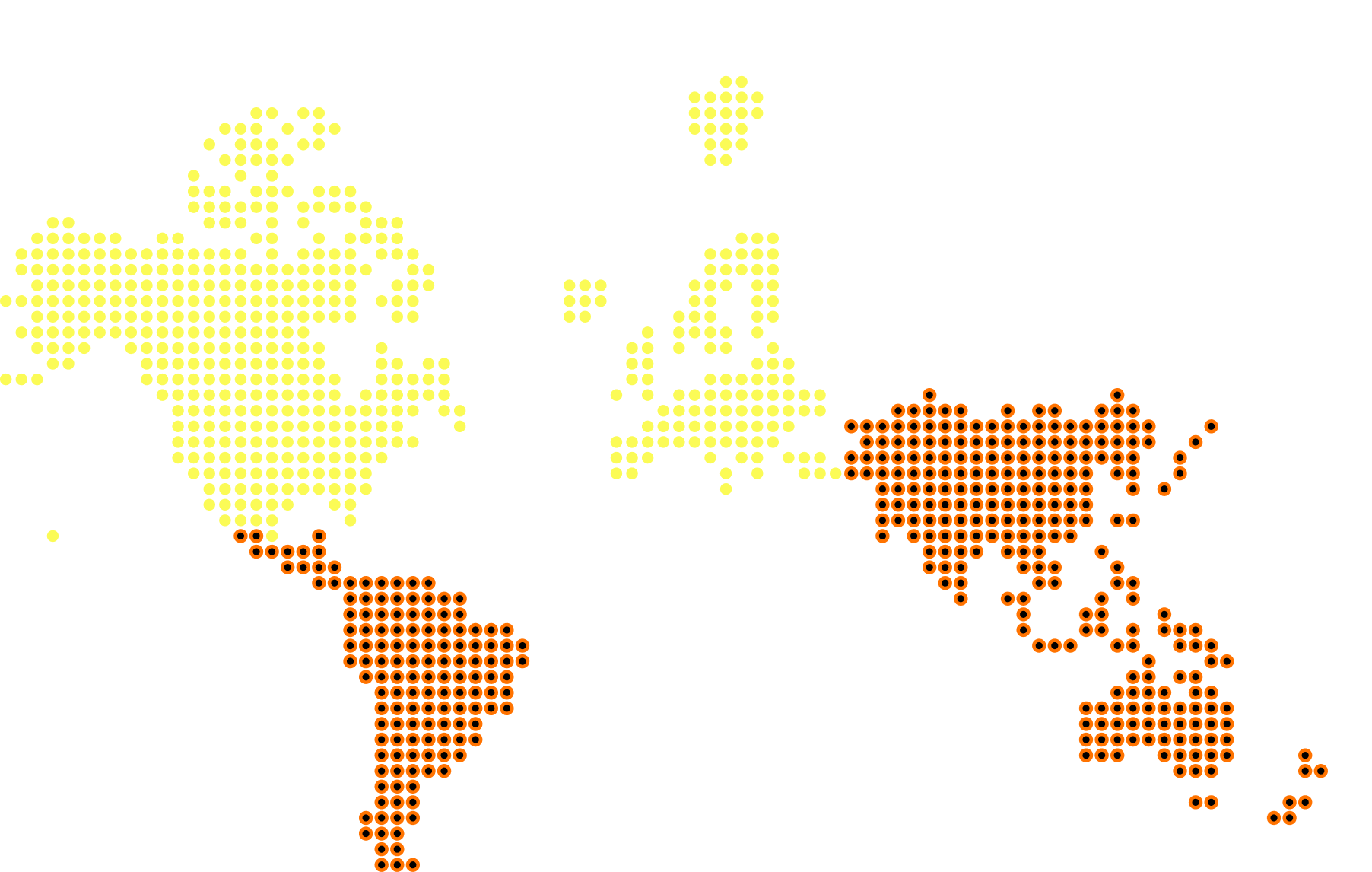
Geographic footprint
Over 160 full-time staff operating across the US, UK, and Europe, with integrated APAC partnerships. Wherever your trial needs to run, the infrastructure is already in place.