DCTs: Reducing study team and participant burden
Decentralised clinical trials offer many benefits such as improved patient access and enhanced recruitment, but they present unique challenges for clinical trial operations teams. The complexities of running decentralised studies and complex participant schedules can place a heavy burden on study teams.
This blog post article will focus on overcoming these challenges and how our approach to unified trial delivery helps reduce study team burden and the time and effort required to manage trials effectively.
Tackling the challenges of decentralised trials
Complex participant schedules
Decentralised trials involve participants from various locations, each with their own schedules and requirements. Coordinating and managing these schedules can be challenging, leading to potential delays and decreased patient satisfaction.
Study team burden
As clinical trials become more complex, the burden on the study teams grows. They face numerous tasks, communication hurdles, and documentation requirements, which can lead to burnout and reduced productivity.
The consequences of not addressing these challenges include delays or failures in clinical trials, poor data quality, and the undermining of trial results' validity.
Our approach to streamlining management and reducing study team burden
At Lindus Health, we understand the complexities faced by clinical trial professionals in managing decentralised trials. Our approach focuses on two core principles:
1. Centralised management
Our unified Citrus platform enables study teams to manage decentralised trials and complex participant schedules seamlessly. With real-time visibility into trial progress, teams can quickly address issues and make data-driven decisions.
2. Streamlined participant and study team flows
Our platform simplifies the management of participant and study team flows, ensuring that all study visits, procedures, and documentation are completed on time. This not only reduces the administrative burden on study teams but also improves patient compliance and data quality.
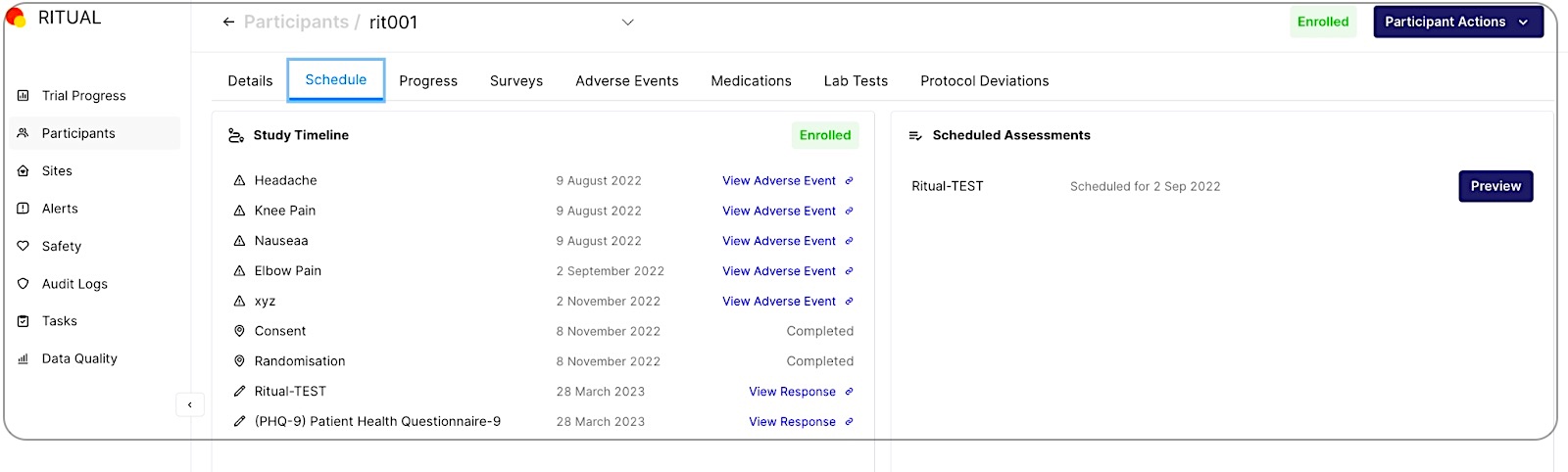
As seen in the screenshot of our Citrus platform below, the site and study team can see all upcoming assessments for trial participants.
Conclusion
Decentralised clinical trials offer significant advantages, but they also introduce new challenges. By centralising management and streamlining participant and study team flows, Lindus Health supports health and biotech pioneers to overcome these hurdles and achieve on-time and on-budget study completion.
Get in touch to learn how Lindus Health can accelerate your next clinical trial.
.png)