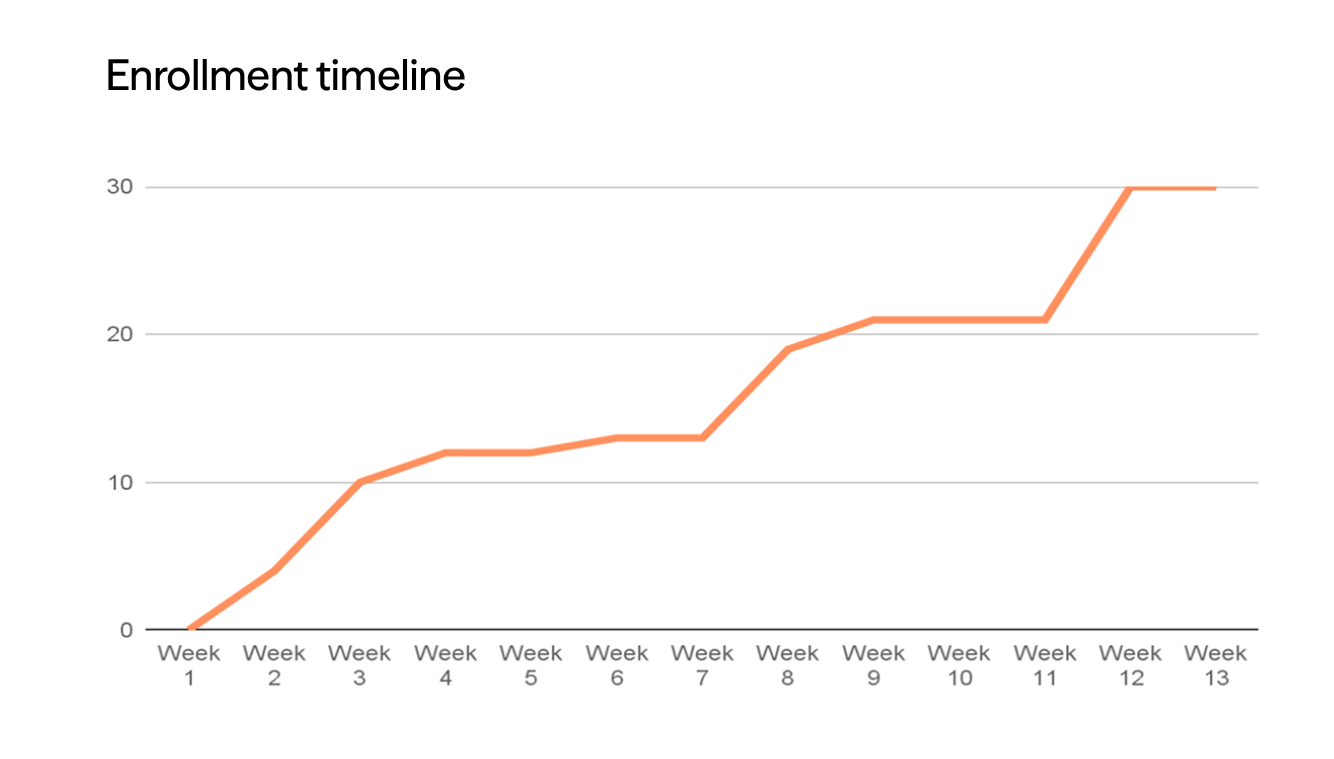
Protocol-mandated washouts trigger severe itch rebound and sleep disruption in patients who have achieved disease control. Anticipating visible symptom relapse, patients withdraw consent or abandon screening. This steadily depletes treatment-naive pools, elevates screen failure rates, and skews enrolled cohorts toward milder phenotypes with lower rebound risk.
Lindus queries 40M+ EHR records to identify patients whose prescribing history suggests washout tolerance before site activation, then deploys patient concierge coordinators who manage scheduling, transport, and check-ins throughout the washout window: sustaining attendance through the highest-attrition phase of your screening funnel.